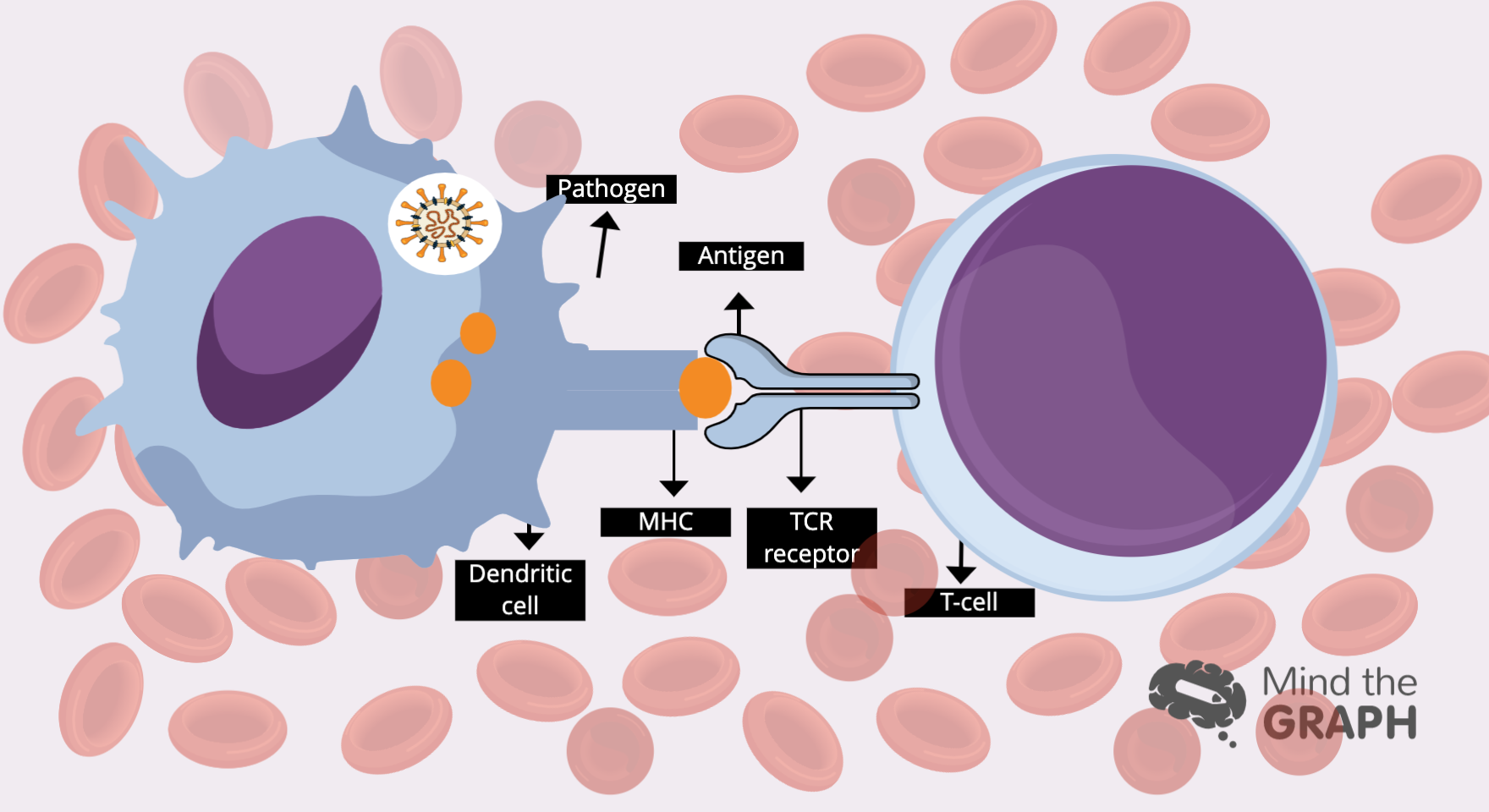
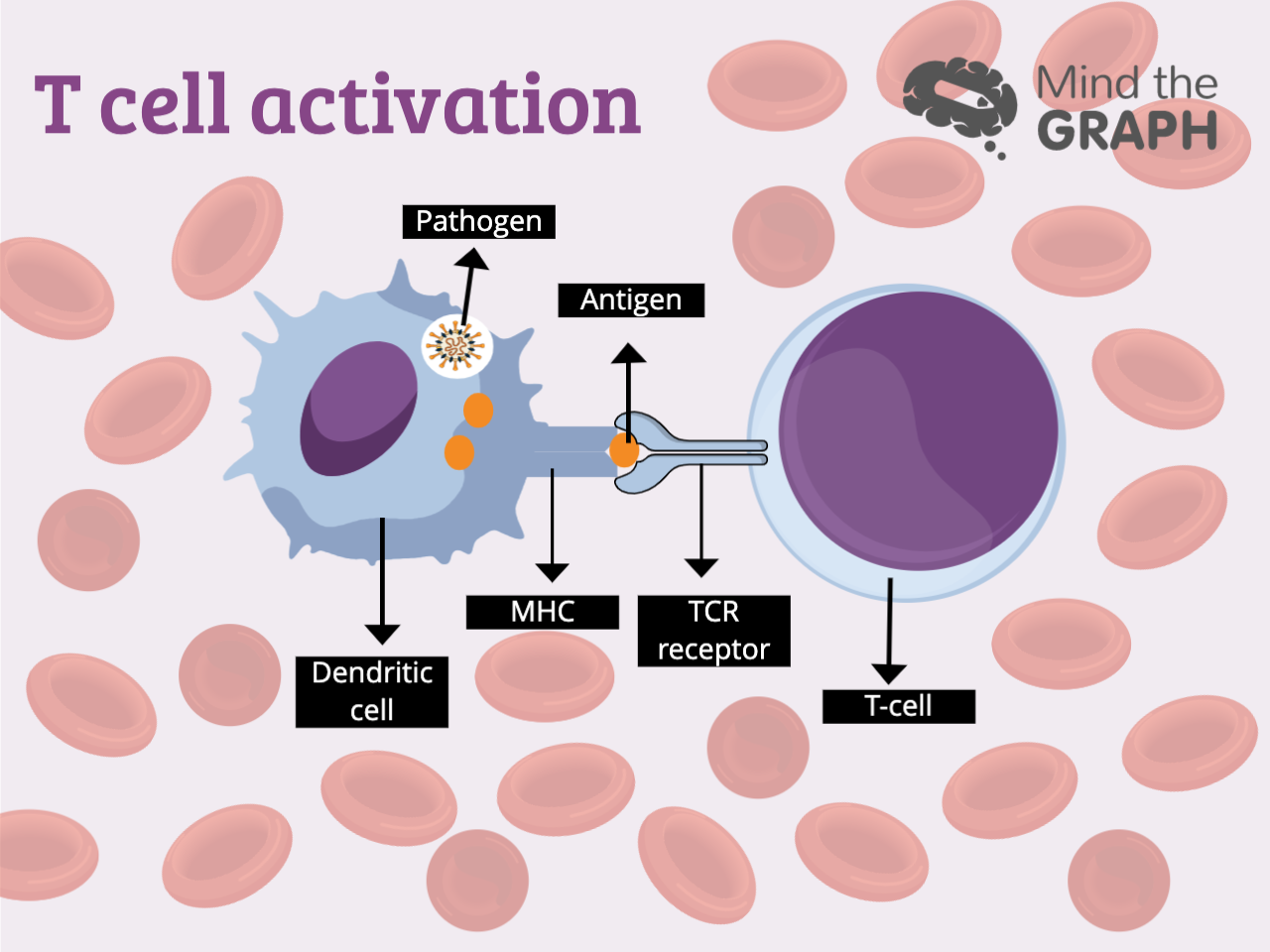
Os glóbulos brancos são responsáveis por defender nosso corpo contra ameaças, mantendo-nos vivos. Mas como essas células identificam uma ameaça? Dentro do corpo humano, com trilhões de células, elas precisam ser específicas e precisas. Caso contrário, eles começam a nos atacar por dentro. Essa responsabilidade recai sobre os ombros das células T, agentes especializados em identificar antígenos nocivos e marcá-los para serem destruídos. Uma das maiores questões sobre a sinalização das células imunes é como as células T distinguem nossas próprias células saudáveis e o que é prejudicial para nós.
Durante o desenvolvimento, o corpo produz milhões de células T com diferentes tipos de receptores aleatoriamente. Afinal, nosso corpo não sabe exatamente o que o futuro reserva. Mas há um problema com essa estratégia. Produzimos células com receptores que identificam peptídeos que pertencem ao corpo. Durante o desenvolvimento, eliminamos a maioria das células T que reagem a essas moléculas “próprias”. No entanto, alguns deles continuam circulando ao longo da vida, protegendo contra células infectadas e anormais sem prejudicar o organismo.
O que impede as células T de marcar nossas células saudáveis?
Um novo artigo intitulado Controle optogenético mostra que a revisão cinética regula a atividade do receptor de células T traz novas evidências para essa questão. Weiner, um dos autores de um dos artigos, disse à Quanta Magazine que “ a célula poderia ter alguma maneira de tomar diferenças muito, muito pequenas na duração da ligação do receptor e amplificar isso para uma resposta celular muito maior”.
A chave para a sinalização das células imunes é o tempo. As substâncias que se ligam aos receptores das células T por menos de cinco segundos são consideradas seguras, enquanto as moléculas de ligação mais longa devem ser destruídas.
Essa teoria, conhecida como revisão cinética, não é nova. Desde a década de 90 alguns pesquisadores propuseram o tempo como chave para a identificação. No entanto, os testes da hipótese encontraram correlações sugestivas entre o tempo de ligação e a ativação das células T, exatamente como a revisão cinética faria, mas esses métodos de teste não conseguiram separar o efeito do tempo dos efeitos de outros fatores biofísicos, como estruturas mudanças nas moléculas e diferenças na força de ligação. Pior, os pesquisadores sabiam que muitos parâmetros potencialmente importantes em jogo que eles não podiam medir ou sequer haviam considerado.
Novos dados optogenéticos na sinalização de células imunes
A optogenética é um campo de pesquisa que usa a luz para controlar as células. Há cientistas usando o método para controlar as interações proteína-proteína em todos os tipos de estudos. Agora, duas equipes de pesquisa estão usando essa tecnologia para estudar a sinalização de células imunes, uma com sede na Alemanha e outra em São Francisco. Apesar das diferenças entre esses dois estudos, os pesquisadores obtiveram resultados semelhantes: quando um evento de ligação durou mais de cinco segundos, a célula T se tornou ativa, mas não conseguiu fazê-lo por nada menos do que isso.
Por que isso acontece ainda não está claro. Mas os pesquisadores têm uma dica para responder que: ” A partir do momento em que um receptor de célula T se liga a uma molécula, uma série de etapas bioquímicas irreversíveis precisam ocorrer antes que a célula seja ativada. Se a molécula se desprender muito cedo durante essa cascata de sinalização , tudo tem que recomeçar desde o primeiro passo.”.
Como sempre na ciência, uma nova resposta cria várias novas perguntas. Sabemos um pouco mais do que antes, mas ainda temos todo um universo desconhecido para explorar.

Subscribe to our newsletter
Exclusive high quality content about effective visual
communication in science.