Yes, unfortunately, your pet is also at risk of getting COVID-19. Just as we are susceptible to SARS-CoV-2 virus infection, certain species of animals are too. Since animals have similar structural features of cells surface proteins present in humans.
A specific protein present acts like a lock and key system, the key is the virus protein responsible for COVID-19 infection, and the lock is the membrane protein located on the cell surface.
If the key – the virus protein – fits the lock – the receptor protein from the host – then the virus can get inside starting its cycle of multiplication causing the host infection.
A study published on the PLOS Computational Biology, used a mathematical model to predict which animals’ species have the necessary features of the host protein that could allow the virus spike protein binding.
Researchers reported some cases around the globe of domestic, farm, and zoo animals testing positive for COVID-19 infection.
Cats, minks, lions, and tigers are some species reported as infected.
At first, studies showed that cats, civets, and ferrets were susceptible to the SARS-CoV-2, while pigs, chickens, and ducks were not.
In a second moment, different results emerged, classifying guinea pigs, mice, and rats as not susceptible, while camels, cattle, horses, sheep, and rabbits were classified as susceptible to SARS-CoV-2 infection.
We should remember that the SARS-CoV-2 virus was first transmitted to humans through an intermediate host.
The two major suspects animals are bats and pangolins. If it is the first time you heard about pangolins, they are nocturnal mammals covered with keratin scales that live in hollow trees and burrows.
Here is an illustration of a pangolin:
To start the infection, the first step the virus has to accomplish is to bind its viral spike protein to the receptor protein from the host cell membrane.
The host receptor protein is an extracellular protease domain of angiotensin-converting enzyme 2, usually known as ACE2.
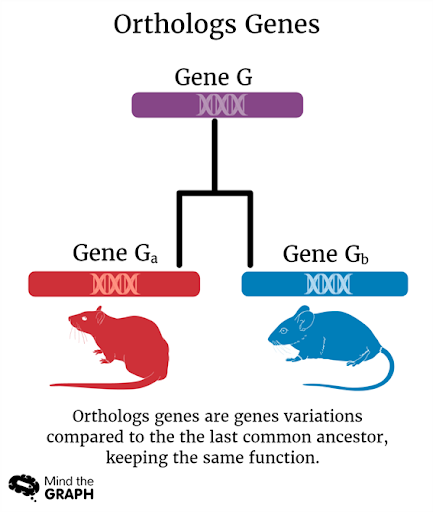
The ACE2 is a single-pass transmembrane protein present on the surface of several tissues. ACE2 doesn’t have only a single structure, it can have multiple structures cross-species while maintaining the same function.
In fact, the ACE2 is classified as an ortholog gene, that is, a gene present in different species that originated from a common ancestral gene.
These structural differences are apparently the reason why the virus SARS-CoV-2 can bind to the ACE2 of some species and not others.
Once the viral-spike-protein is connected to the ACE2, the virus gets inside the cell and starts using the host cellular machinery to multiply leading to infection.
As a result, having a better understanding about ACE2 receptor would certainly lead to a more precise explanation of why some animal’s species are susceptible to infection while others are not.
In addition, it would help scientists in drug development too, applying antiviral strategies, like producing fake lockers for the virus to bind instead of the actual locker from the host.
As well as to better track host intermediate animals for future virus infections.
So, characterizing the three-dimensional structure of the ACE2 and its interactions at the atomic level using computational modeling, was the goal of Rodrigues and colleagues work.
This type of characterizing study is experimentally expensive and not applicable on large scale turning difficult to reach conclusive data, meanwhile, computation modeling is fast and inexpensive, being very beneficial to fill some gaps in structural framework knowledge.
The study, took 28 animal species for analysis, generating a 3D model of the ACE2 bonded to the viral spike protein.
The results showed that there are key locations in the ACE2 amino acid sequence, where due to mutations the viral protein could not bind to the host cell.
Specifically, the ACE2 sequence lack one amino acidic responsible for an intermolecular salt-bridge observed between ACE2 and the binding site of the viral protein, as reported by Rodrigues and his team.
This result is supported by other published studies, which have demonstrated experimentally that this mutant lack is the absence of negatively-charged amino acid, enabling the viral protein to bound.
The study also indicates that the SARS-CoV-2 infection is more complex than previously thought, with more than one way to infect the host.
Thus, it is important to clarify that reducing the ACE2 availability can decrease the chances of infection; however, that does not prevent the infection to occur.
And that the computation models cannot be used alone to predict species risks of infection – additional experimental data is necessary to have a more reliable conclusion.
So, I guess we must continue following the pandemic protocols and do what we have been already doing… Using masks, keeping social distance as much as possible, not going to indoors and crowded spaces, for our safety and especially our beloved pets, ourselves, and our family.
This fluffy cat is so beautiful, isn’t it? You can find this and many others animal illustrations in Mind the Graph platform.
Veterinary, ornithology, entomology, and zoology illustrations are also available on the platform to be used in your infographic. Start using Mind the Graph right now!

Subscribe to our newsletter
Exclusive high quality content about effective visual
communication in science.